
Selected by Science as one of the three topline images on the homepage.
It is well known that protonated water forms when an acid dissolves in water, with the characteristic of hydronium ion or hydrated proton (H3O+). To date, the existence of the hydrated proton has already been realized at the end of 19th century by Swedish physicist/chemist Svante Arrhenius and German chemist Wilhelm Ostwald. Hydrated protons are ubiquitous in solutions and involved in a variety of physical, chemical, biological, and energy-related processes. In particular, the solvation and dynamics of hydrated protons at water-solid interfaces are highly relevant to the key features of electrochemical reactions, proton channels and hydrogen-fuel cells, but their nature has been under longstanding and lively debate.
Many different forms of hydrated protons in water have been reported, among which Eigen cation (H3O+(H2O)3) and Zundel cation (H5O2+) are the most representative ones (Figure 1). However, the configuration of Zundel and Eigen cations has been experimentally elusive, especially at the interfaces, mainly due to the lack of atomic-level characterization. Moreover, imaging and identifying hydrated protons within the H-bonding network of water remains a great challenge due to the high similarity between the hydronium (H3O+) and the water molecule (H2O).
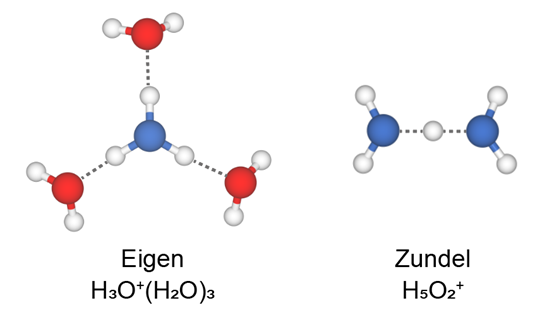
Figure 1 The structural models of Eigen and Zundel cations. Eigen cation: one flat hydronium ion at the center is H-bonded with three water molecules; Zundel cation: two water molecules share an extra H+ in a symmetric H-bonding configuration.
Now, the team led by Prof. Ying Jiang, Prof. Ji Chen and Prof. Enge Wang from International Center for Quantum Materials (ICQM), School of Physics of Peking University, in collaboration with Prof. Jing Guo from College of Chemistry of Beijing Normal University, successfully visualize the atomic structures of stable Eigen/Zundel cations on metal surfaces and reveal a new proton transfer mechanism at the water-solid interfaces, through a combined study using qPlus-based atomic force microscope (AFM), path integral molecular dynamics (PIMD) simulations and density functional theory (DFT) calculations. This work was published in Science on July 15th, 2022.
In this work, the researchers performed the coadsorption of deuterium (D) atoms and D2O molecules on different metal substrates (Au, Cu, Pt, Ru), where the D atoms are ionized by transferring electrons to the metal substrate. The ionized D+ and D2O molecules can self-assemble into a two-dimensional (2D) hydronium-water overlayer. The researchers have substantially improved the resolution and sensitivity of the qPlus-AFM, such that the individual hydronium ions (D3O+) solvated in the water network could be directly visualized. At low D coverages, the hydronium-water overlayer could be considered as the self-assembly of Eigen cations (D9O4+) (Figure 2A), where one flat hydronium ion at the center is H-bonded with three water molecules.
Interestingly, the researchers observed the conversion from Eigen cations to Zundel cations when dosing D at higher coverages. Using the qPlus-AFM, the Zundel cation could be directly visualized as a dimer-like structure with a depression feature at the center (Figure 2B), which is attributed to the two flat water molecules with a shared D+ in a symmetric H-bonding configuration. PIMD simulations revealed that the formation and stabilization of the Zundel cation was closely related to the nuclear quantum effects (NQEs), which significantly promoted the proton delocalization between the water molecules and resulted in the formation of symmetric H-bond.
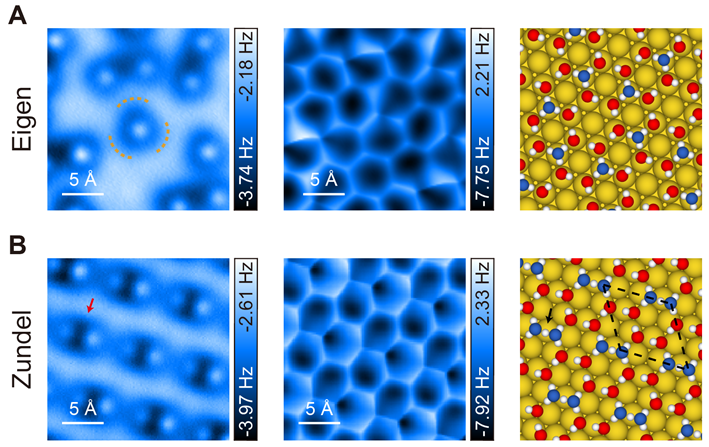
Figure 2 High-resolution AFM images and atomic structures of self-assembled Eigen (A)/Zundel (B) water monolayer on Au(111) surface. O atoms in the hydronium ions and water molecules are denoted as blue and red spheres, respectively.
Furthermore, the researchers performed the tip manipulation experiments to explore the dynamics of hydronium ions at the interface and found that two Eigen cations could combine into one Zundel cation, accompanied with a simultaneous proton transfer from the water layer to the substrate, resulting in a stable Zundel+D* configuration (Figure 3A-C). Such a process (from two Eigens to Zundel+D*) actually corresponds to a new proton transfer pathway, which is beyond the previously known elementary steps of hydrogen evolution reaction (HER).
Moreover, the researchers found that the Zundel was always preferred on Pt(111), while there was a clear transition from Eigen to Zundel structure on Au(111) (Figure 3D). It suggests that distant hydrated protons were more inclined to combine on Pt(111), thus facilitating the production of H2. At low Zundel density, the Zundel+H* configuration on Pt(111) may allow efficient H2 evolution through the Heyrovsky reaction pathway (H+ + e- + H* → H2). However, at high Zundel density, the coverage of H* on the Pt(111) is also increased, which opens up the Tafel reaction pathway (2H* → H2). This feature may provide new insights into the behavior of reaction kinetics on Pt(111) and help to boost the HER efficiency from a new perspective.
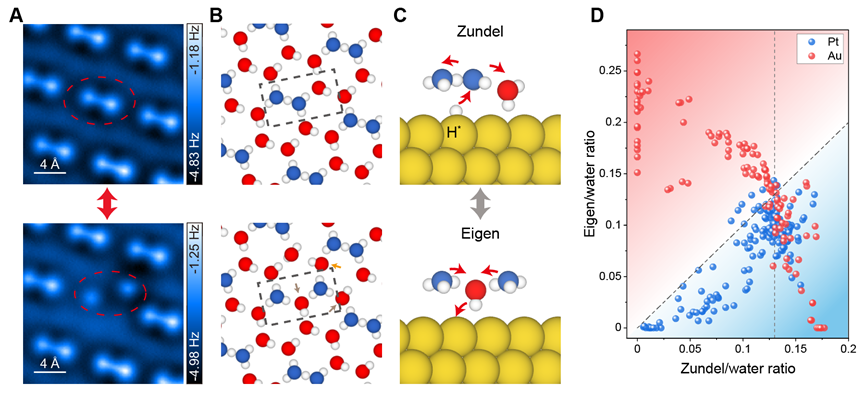
Figure 3 (A, B) AFM images (A) and the schematic H-bonding arrangements (B) of the ordered Zundel cations and the tip-induced two Eigen cations in the hydronium-water layer. (C) Schematic models of the interconversion between the Zundel and Eigen cations. (D) Density correlation between the Eigen and Zundel cations on the Au(111) and Pt(111) surfaces.
This work represents the first atomically resolved experiment towards understanding the solvation and dynamics of hydrated protons at interfaces. It suggests that the Zundel and Eigen cations, which are transient in bulk water, can have exceptionally long lifetimes at water-solid interfaces. In addition, the stabilization of 2D Zundel-water layer with long-range order may also open up new possibility of exploring novel physical properties related to symmetric H bonds in the absence of high pressure, such as superconductivity, phase transition, ultrafast proton transfer and superionic.
Dr. Ye tian, Jiani Hong, Dr. Sifan You from ICQM of Peking University and Dr. Duanyun Cao from School of Materials Science and Engineering, Beijing Institute of Technology are the co-first authors of this work. Prof. Ying Jiang, Prof. Enge Wang, Prof. Ji Chen and Prof. Jing Guo are the co-corresponding authors. This work received supports from National Natural Science Foundation of China, Ministry of Science and Technology of China, Chinese Academy of Sciences, and Collaborative Innovation Center of Quantum Matter.
Paper link:
Y. Tian, J. Hong, D. Cao, S. You, Y. Song, B. Cheng, Z. Wang, D. Guan, X. Liu, Z. Zhao, X.-Z. Li, L.-M. Xu, J. Guo*, J. Chen*, E.-G. Wang* and Y. Jiang*, Visualizing Eigen/Zundel cations and their interconversion in monolayer water on metal surfaces, Science 377, 315-319 (2022). (https://www.science.org/doi/10.1126/science.abo0823)
